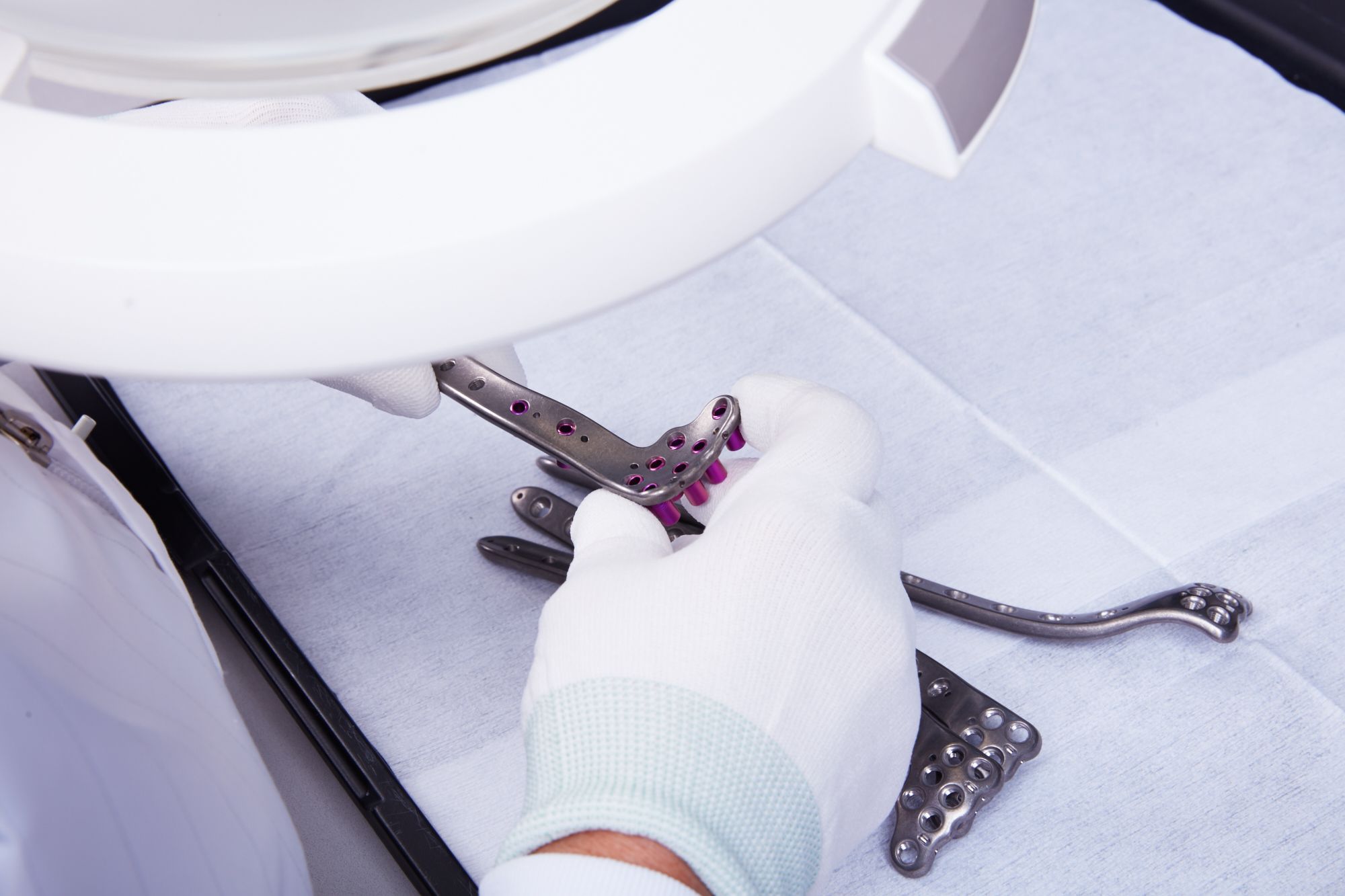
Quality
We are dedicated to delivering reliable and safe implants to our customers
Our team of experts ensures that all of our manufacturing processes are compliant with ISO 13485 and FDA regulations. With our state-of-the-art equipment and rigorous quality control measures, we are dedicated to delivering reliable and safe medical devices to our customers. We take pride in our quality-first approach and strive to exceed our customers' expectations every step of the way.
Quality capabilities
Our commitment to quality is evident in everything we do at SpiTrex Orthopedics. As an ISO 13485 certified and FDA registered medical device contract manufacturer, we ensure that all of our processes and procedures meet the highest standards of quality and compliance.
ISO 13485 certified
Our ISO 13485 certification ensures that we have implemented a quality management system that meets regulatory requirements and that we consistently provide products and services that meet customer and regulatory requirements.
FDA registered
As an FDA registered facility, we have undergone rigorous inspections and audits to demonstrate compliance with FDA regulations and guidelines.
Validated equipment and cleaning processes
We use validated equipment and cleaning processes to ensure that our manufacturing environment and equipment meet the required cleanliness standards.
CMM and vision system inspection
We use state-of-the-art inspection equipment, including Coordinate Measuring Machines (CMM) and Vision Systems, to verify the accuracy and quality of our manufactured components.
First article inspection
Our First Article Inspection (FAI) process is a key part of our quality control program. We perform FAIs on all new parts to ensure that they meet customer requirements and specifications.
At Spitrex Orthopedics, we hold ourselves to the highest standards of quality throughout our operations. We are committed to providing our customers with the highest quality products and services, and we continuously strive to improve our processes and procedures to ensure that we exceed expectations.

